|
Figure 21.7 "Isolating Lithium from Spodumene, a Lithium Silicate Ore" illustrates the isolation of liquid lithium from a lithium silicate ore by this process. The steps in this process are leaching, which uses sulfuric acid to dissolve the desired alkali metal ion and Al 3+ from the ore basic precipitation to remove Al 3+ from the mixture as Al(OH) 3 selective precipitation of the insoluble alkali metal carbonate dissolution of the salt again in hydrochloric acid and isolation of the metal by evaporation and electrolysis. Rubidium is obtained commercially by isolating the 2%–4% of Rb present as an impurity in micas, minerals that are composed of sheets of complex hydrated potassium–aluminum silicates.Īlkali metals are recovered from silicate ores in a multistep process that takes advantage of the pH-dependent solubility of selected salts of each metal ion. No concentrated sources of rubidium are known, for example, even though it is the 16th most abundant element on Earth. The other alkali metals are found in low concentrations in a wide variety of minerals, but ores that contain high concentrations of these elements are relatively rare. Massive deposits of essentially pure NaCl and KCl are found in nature and are the major sources of sodium and potassium. All the isotopes of Fr have very short half-lives, in contrast to the other elements in group 1.Įquation 21.6 2RbOH(s) + Mg(s) → 2Rb(l) + Mg(OH) 2(s) Francium (Fr) is found in only trace amounts in nature, so our knowledge of its chemistry is limited. Known to chemistry students as the inventor of the Bunsen burner, Bunsen’s spectroscopic studies of ores showed sky blue and deep red emission lines that he attributed to two new elements, Cs and Rb, respectively. Cesium (Cs) and rubidium (Rb) were not discovered until the 1860s, when Robert Bunsen conducted a systematic search for new elements. (The ashes produced by the combustion of wood are largely composed of potassium and sodium carbonate.) Lithium (Li) was discovered 10 years later when the Swedish chemist Johan Arfwedson was studying the composition of a new Brazilian mineral. For these reasons, the group 1 elements were unknown until the early 19th century, when Sir Humphry Davy first prepared sodium (Na) and potassium (K) by passing an electric current through molten alkalis. Although some of their ores are abundant, isolating them from their ores is somewhat difficult. 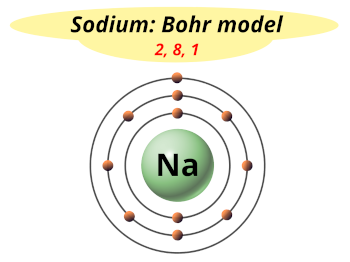
The alkali metals are so reactive that they are never found in nature in elemental form. To be familiar with the reactions, compounds, and complexes of the alkali metals.To describe how the alkali metals are isolated.These examples are programmatically compiled from various online sources to illustrate current usage of the word 'sodium.' Any opinions expressed in the examples do not represent those of Merriam-Webster or its editors. Paige Grandjean, Southern Living, 13 Oct. 2023 There is plenty of salt from the ham, no need to add more, and unsalted chicken stock is hard to find unless making it yourself (low sodium, yes, but that is still too much salt by the time the soup is finished). 2023 The group fabricated their device using CFRP, sodium potassium niobate (KNN) nanoparticles, and epoxy resin. 2023 Qi and his research team think the findings could support interventions to help people lower their sodium intake and, perhaps, lower their diabetes risk. 2023 So the ability to build a really sustainable setup with sodium ion is definitely there.  
Alison Klesman, Discover Magazine, 11 Nov. 2023 The data show evidence of carbon dioxide in the atmosphere other telescopes have found water vapor, sodium and potassium. 2023 Among them is a reactor that was cooled by sodium instead of water and operated for 30 years in Idaho. Andrea Wurzburger, Better Homes & Gardens, 22 Nov. 2023 Water softeners use a process called ion exchange in order to replace those minerals with sodium, making the water less hard. 
Recent Examples on the Web Even with the recent slump in lithium prices, sodium is still a cheaper option.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
